| CAS NO: | 1072833-77-2 |
| 规格: | ≥98% |
| 包装 | 价格(元) |
| 5mg | 电议 |
| 10mg | 电议 |
| 25mg | 电议 |
| 50mg | 电议 |
| 100mg | 电议 |
| 250mg | 电议 |
| 500mg | 电议 |
| Molecular Weight (MW) | 361.03 |
|---|---|
| Formula | C14H19BCl2N2O4 |
| CAS No. | 1072833-77-2 |
| Storage | -20℃ for 3 years in powder form |
| -80℃ for 2 years in solvent | |
| Solubility (In vitro) | DMSO: 72 mg/mL (199.4 mM) |
| Water: <1 mg/mL | |
| Ethanol: 9 mg/mL (24.9 mM) | |
| Solubility (In vivo) | 0.5% hydroxyethyl cellulose: 30 mg/mL |
| Synonyms | MLN-2238; MLN2238; IXAZOMIB; MLN 2238; Trade name: Ninlaro. IUPAC/Chemical Name: (R)-(1-(2-(2,5-dichlorobenzamido)acetamido)-3-methylbutyl)boronic acid InChi Key: MXAYKZJJDUDWDS-LBPRGKRZSA-N InChi Code: InChI=1S/C14H19BCl2N2O4/c1-8(2)5-12(15(22)23)19-13(20)7-18-14(21)10-6-9(16)3-4-11(10)17/h3-4,6,8,12,22-23H,5,7H2,1-2H3,(H,18,21)(H,19,20)/t12-/m0/s1 SMILES Code: CC(C)C[C@@H](B(O)O)NC(CNC(C1=CC(Cl)=CC=C1Cl)=O)=O |
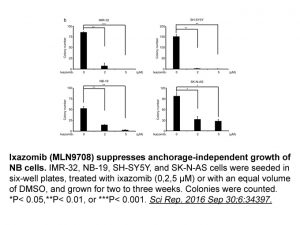
| In Vitro | In vitro activity: At higher concentrations, MLN2238 also inhibits the caspase-like (β1) and trypsin-like (β2) proteolytic sites with IC50 of 31nM and 3.5uM, respectively. MLN2238 inhibits Calu-6 cell with IC50 of 9.7 nM. MLN2238 is a selective, potent, and reversible inhibitor of the proteasome in tumor cells. MLN2238 shows time-dependent reversible proteasome inhibition. Both MLN2238 and Bortezomib shows time-dependent reversible proteasome inhibition; however, the proteasome dissociation half-life for MLN2238 is determined to be ~6-fold faster than that of Bortezomib (18 and 110 minutes, respectively). MLN2238 dissociates more rapidly from the proteasome than Bortezomib, consistent with faster recovery of proteasome activity observed in the Proteasome-Glo assay. MLN2238 has a greater overall tumor pharmacodynamic effect than Bortezomib as assessed by 20S inhibition. MLN2238 is the biologically active form of MLN9708. Kinase Assay: Calu-6 cells are cultured in MEM containing 10% fetal bovine serum and 1% penicillin/streptomycin and plated 1 day before the start of the experiment at 1 × 104 cells per well in a 384-well plate. Proteasome activity is assessed by monitoring hydrolysis of the chymotrypsin-like substrate Suc-LLVY-aminoluciferin in the presence of luciferase using the Proteasome-Glo assay reagents according to the manufacturer's instructions. Luminescence is measured using a LEADseeker instrument. Cell Assay: Calu-6 cells are cultured in MEM containing 10% FBS and 1% penicillin/streptomycin and plated 1 day before the start of the experiment at 1 × 104 cells per well in a 384-well plate. For IC50 determinations, cells are treated with varying concentrations of Bortezomib or MLN2238 in DMSO (0.5% final, v/v) for 1 hour at 37 °C. For reversibility experiments, cells are treated with either 1 μM Bortezomib or MLN2238 for 30 minutes at 37 °C and then washed thrice in medium to remove the Bortezomib or MLN2238. Cells are incubated for an additional 4 hours at 37 °C, after which the medium is removed and replaced with fresh medium. |
|---|---|
| In Vivo | MLN2238 induces a greater pharmacodynamic response than Bortezomib in xenograft tumors. MLN2238 shows greater maximum and sustained tumor proteasome inhibition compared with Bortezomib in xenograft models. These results confirm that the improved tumor exposure seen with MLN2238 translates into an improved tumor pharmacodynamic response both at the level of and downstream from the proteasome. MLN2238 shows antitumor activity in the CWR22 xenograft model. MLN2238 shows greater tumor pharmacodynamic responses in WSU-DLCL2 xenografts compared with Bortezomib. Similarly, Bortezomib treatment only led to a minor increase in GADD34 levels in WSU-DLCL2 xenograft tumors, whereas MLN2238 strongly induces its expression. MLN2238 has an improved pharmacodynamic profile and antitumor activity compared with Bortezomib in both OCI-Ly10 and PHTX22L models |
| Animal model | CB-17 SCID mice are subcutaneously inoculated with MM.1S cells |
| Formulation & Dosage | Dissolved in 5% 2-hydroxypropyl-β-cyclodextrin; 11 mg/kg; i.v. injection |
| References | Cancer Res. 2010 Mar 1;70(5):1970-80; Clin Cancer Res. 2011 Dec 1;17(23):7313-23. |
|
|
|
|
|
|  |
